Indications
Rehabilitation & Sports

Intimate Health
* Not under Notified Body certification. These indications are not in the scope of medical device regulation (EU) 2017/745.

* Not under Notified Body certification. These indications are not in the scope of medical device regulation (EU) 2017/745.

COMPACT Plus incorporates INDIBA’s latest innovations, offering superior precision and advanced treatment efficacy, while maintaining ease of use for professionals.





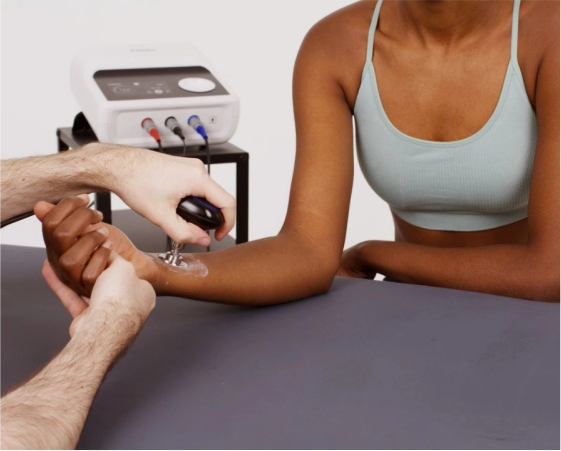
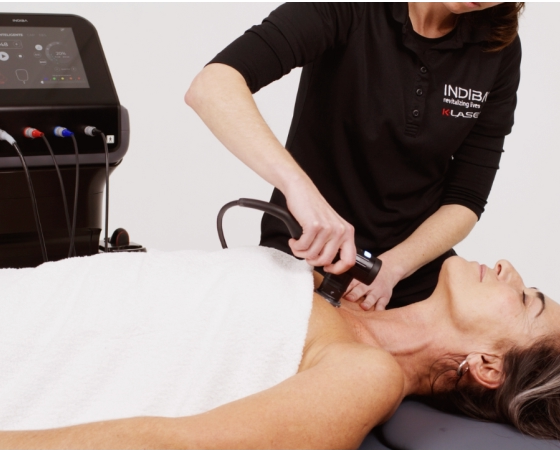


Precision-engineered stainless steel electrodes designed for deep myofascial release, improving tissue elasticity and circulation.

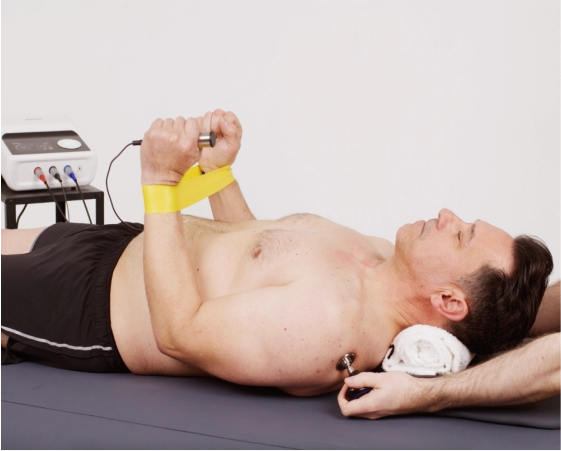
Enables physiotherapists to provide RF therapy while patients perform active rehabilitation exercises.

Specialized capacitive intracavitary electrodes tailored for pelvic floor and intimate wellness treatments, ensuring precision and comfort.

Available in multiple sizes, for external and intracavitary applications, allowing professionals to customize treatments for musculoskeletal and pelvic health applications.
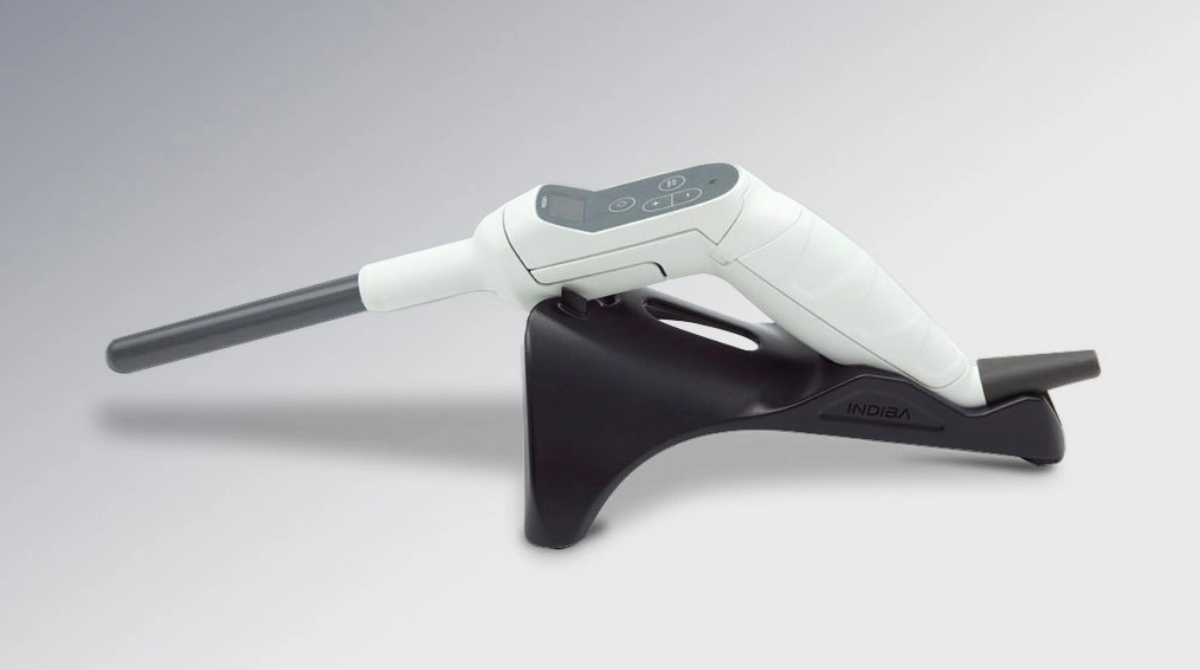
A revolutionary intracavitary handle for gynecological treatments, offering precision, safety, and fast, effective results with advanced temperature control and single-use CAP electrodes.
Oncological patients Oncological patients/consumers:
1. Do not use INDIBA directly on the tumor or on regional lymph nodes that may be affected by the tumor.
2. Before treating a patient/consumer undergoing oncological treatment with INDIBA, consult the oncologist.
3. Patients/consumers considered to be in tumor remission can be safely treated.
Consult the doctor in charge.
Apply less power and pay attention to changes in skin colour. Palpate the skin to check local temperature. Diabetic patients/consumers, even if pathology is ontrolled, are to be considered in this group.
Avoid aesthetic treatments around the area.
Avoid treatments with high temperatures near prominent bones.
It must be performed without temperature.
Consult an expert on the suitability of the time between treatments.